
At slightly above the critical temperature (310 K), in the vicinity of the critical pressure, the line is almost vertical. Thus, above the critical temperature a gas cannot be liquefied by pressure. At the critical point, (304.1 K and 7.38 MPa (73.8 bar)), there is no difference in density, and the 2 phases become one fluid phase. As the critical temperature is approached (300 K), the density of the gas at equilibrium becomes higher, and that of the liquid lower. The system consists of 2 phases in equilibrium, a dense liquid and a low density gas. At well below the critical temperature, e.g., 280 K, as the pressure increases, the gas compresses and eventually (at just over 40 bar) condenses into a much denser liquid, resulting in the discontinuity in the line (vertical dotted line). The appearance of a single phase can also be observed in the density-pressure phase diagram for carbon dioxide (Fig. 1) the boiling curve separates the gas and liquid region and ends in the critical point, where the liquid and gas phases disappear to become a single supercritical phase. In the pressure-temperature phase diagram (Fig. Carbon dioxide density-pressure phase diagramįigures 1 and 2 show two-dimensional projections of a phase diagram. The critical point of a binary mixture can be estimated as the arithmetic mean of the critical temperatures and pressures of the two components,įigure 2. This behavior has been found for example in the systems N 2-NH 3, NH 3-CH 4, SO 2-N 2 and n-butane-H 2O. 
However, exceptions are known in systems where one component is much more volatile than the other, which in some cases form two immiscible gas phases at high pressure and temperatures above the component critical points. Typically, supercritical fluids are completely miscible with each other, so that a binary mixture forms a single gaseous phase if the critical point of the mixture is exceeded. Therefore, close to the critical temperature, solubility often drops with increasing temperature, then rises again. 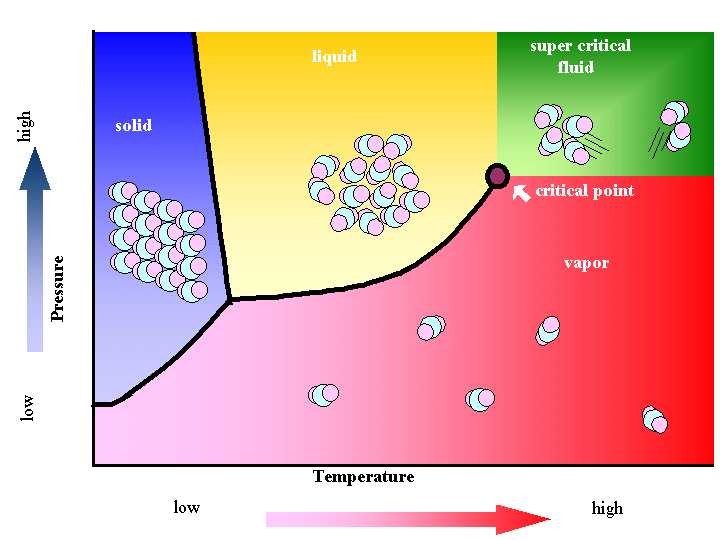
However, close to the critical point, the density can drop sharply with a slight increase in temperature. At constant density, solubility will increase with temperature. The relationship with temperature is a little more complicated. Since density increases with pressure, solubility tends to increase with pressure. Solubility in a supercritical fluid tends to increase with density of the fluid (at constant temperature). One of the most important properties is the solubility of material in the fluid. By changing the pressure and temperature of the fluid, the properties can be "tuned" to be more liquid-like or more gas-like. Comparison of gases, supercritical fluids and liquids Īlso, there is no surface tension in a supercritical fluid, as there is no liquid/gas phase boundary. Table 2 shows density, diffusivity and viscosity for typical liquids, gases and supercritical fluids. †Source: International Association for Properties of Water and Steam ( IAPWS) Critical properties of various solvents Solvent In Table 1, the critical properties are shown for some substances that are commonly used as supercritical fluids. Supercritical fluids generally have properties between those of a gas and a liquid. This can be used to extract a substance and transport it elsewhere in solution before depositing it in the desired place by simply allowing or inducing a phase transition in the solvent. carbon dioxide) but insoluble in the gaseous or liquid state-or vice versa. An interesting property is that some substances are soluble in the supercritical state of a solvent (e.g. Carbon dioxide and water are the most commonly used supercritical fluids they are often used for decaffeination and power generation, respectively. They are used as a substitute for organic solvents in a range of industrial and laboratory processes. Supercritical water is found on Earth, such as the water issuing from black smokers, a type of underwater hydrothermal vent. Supercritical fluids occur in the atmospheres of the gas giants Jupiter and Saturn, the terrestrial planet Venus, and probably in those of the ice giants Uranus and Neptune. Also, near the critical point, small changes in pressure or temperature result in large changes in density, allowing many properties of a supercritical fluid to be "fine-tuned". SCF are much superior to gases in their ability to dissolve materials like liquids or solids. It can effuse through porous solids like a gas, overcoming the mass transfer limitations that slow liquid transport through such materials. Not to be confused with superfluidity or supercritical flow.Ī supercritical fluid ( SCF) is any substance at a temperature and pressure above its critical point, where distinct liquid and gas phases do not exist, but below the pressure required to compress it into a solid.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
